GLP-1 Gene Therapy: Could Your Body Become Its Own Ozempic Factory?
Audio Overview
GLP-1 Gene Therapy: Could Your Body Become Its Own Ozempic Factory?
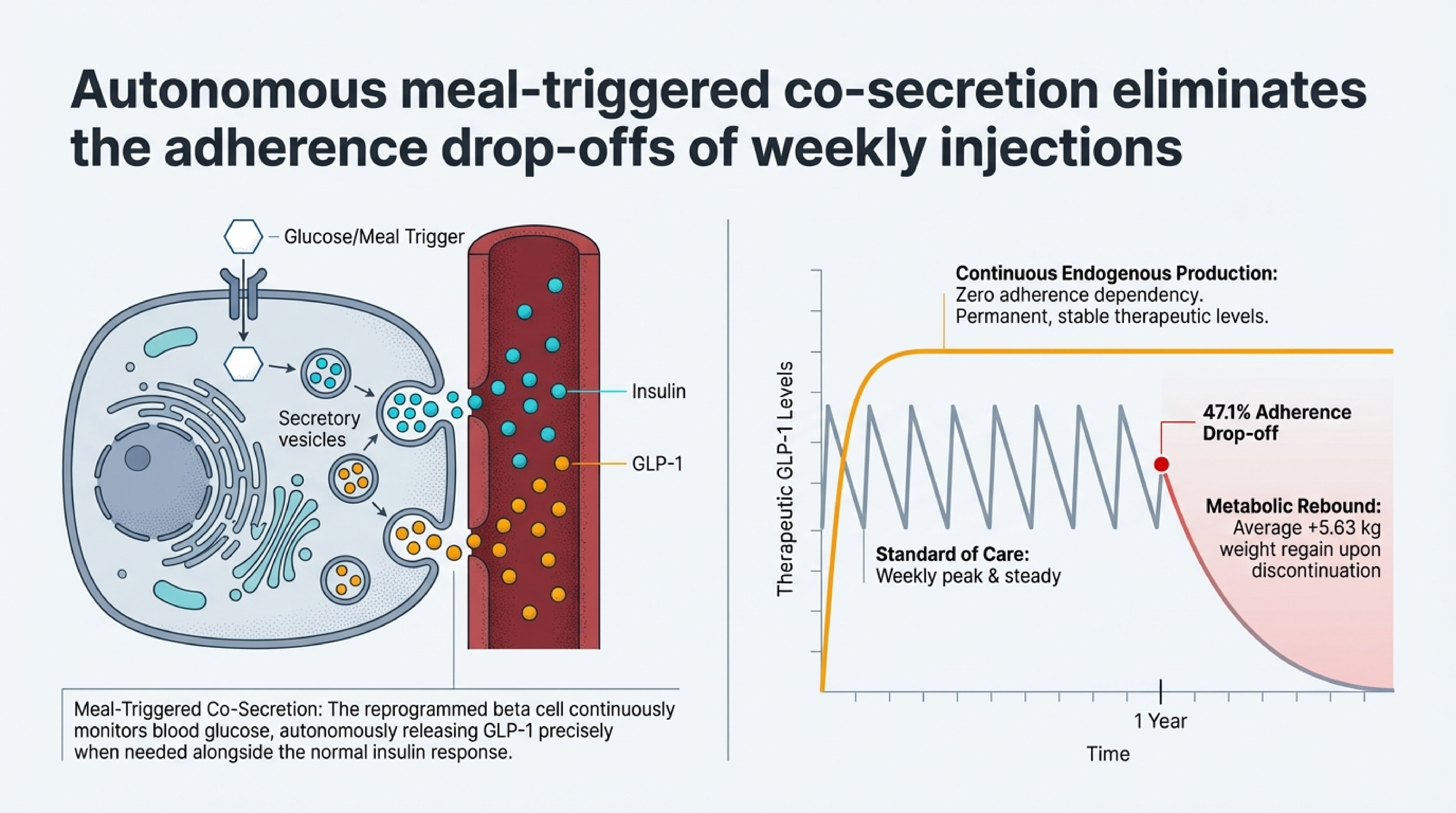
The Adherence Problem Nobody Wants to Talk About

Here's a number that should bother everyone in medicine: only 32 percent of people without diabetes who start a GLP-1 drug are still taking it a year later. That means roughly two out of three patients walk away from a medication that was actually working for them. And when they stop, the weight comes back — a 2025 meta-analysis in The Lancet found that patients who discontinued semaglutide or tirzepatide regained an average of nearly 10 kilograms. The landmark STEP 1 trial extension was even more sobering: two-thirds of the lost weight returned within twelve months of stopping.
I think about this constantly. We finally have drugs that produce 15 to 20 percent body weight loss, that reduce cardiovascular events by 20 percent — the SELECT trial proved that in over 17,000 patients — and that are genuinely bending the national obesity curve downward for the first time in decades. The U.S. obesity rate dropped from 39.9 percent in 2022 to 37 percent in 2025. That's roughly 7.6 million fewer adults classified as obese. GLP-1 adoption more than doubled over the same period, from 5.8 to 12.4 percent of American adults.
And yet we're losing patients. Cost is part of it — over a thousand dollars a month without insurance, and only 19 percent of large employers cover GLP-1s for weight loss. Side effects are another factor. The majority of patients experience gastrointestinal symptoms at some point: nausea in about 42 percent, vomiting in 22 percent. These are manageable with dose titration, but they drive roughly half of early discontinuations. And then there's the psychological weight of committing to weekly injections indefinitely, with no finish line in sight.
So when someone asks me whether GLP-1 gene therapy is real science or just hype, my answer is: this is the most logical next question medicine could be asking right now.
What GLP-1 Gene Therapy Actually Is
Think of it like this. Right now, taking a GLP-1 drug is like hiring someone to come to your house every week and refill your water tank. It works great — as long as they keep showing up. GLP-1 gene therapy is more like connecting your house to a well. One installation, and the water just comes from the ground. Your own cells become the source.
The idea is straightforward in principle: deliver a set of genetic instructions into specific cells so they start producing GLP-1 on their own. Instead of injecting a synthetic version of the hormone from the outside, you reprogram cells to manufacture it internally. One treatment. Potentially permanent.
Two approaches are furthest along, and they differ in how they get those instructions into your cells. Fractyl Health, based in Massachusetts, uses an adeno-associated virus — AAV, which is a modified, non-disease-causing virus that acts as a delivery vehicle — to carry DNA instructions directly into insulin-producing beta cells in the pancreas. RenBio, in New York, takes a different route entirely: they use what they call "naked DNA," a plasmid dissolved in simple saline solution, no viral carrier needed. Each approach has its own tradeoff profile in terms of delivery efficiency, immune response risk, and the ability to re-treat if needed.

Fractyl's Rejuva Platform: From Mice to Human Trials
Here's where this story gets fascinating. When the original reporting on GLP-1 gene therapy circulated in late 2025, Fractyl was still primarily discussing rodent data. That's no longer the case.
The company's program — now called Rejuva, no longer GLP-1 PGTX — has two candidates in development. RJVA-001 targets type 2 diabetes with GLP-1 alone. RJVA-002 targets obesity with a dual GIP/GLP-1 construct, encoding both hormones under an engineered insulin promoter. In preclinical obesity models, RJVA-002 produced approximately 30 percent weight loss after a single dose — and the weight loss trajectory hadn't plateaued at five weeks. That's a meaningful jump from the earlier 20 percent figure.
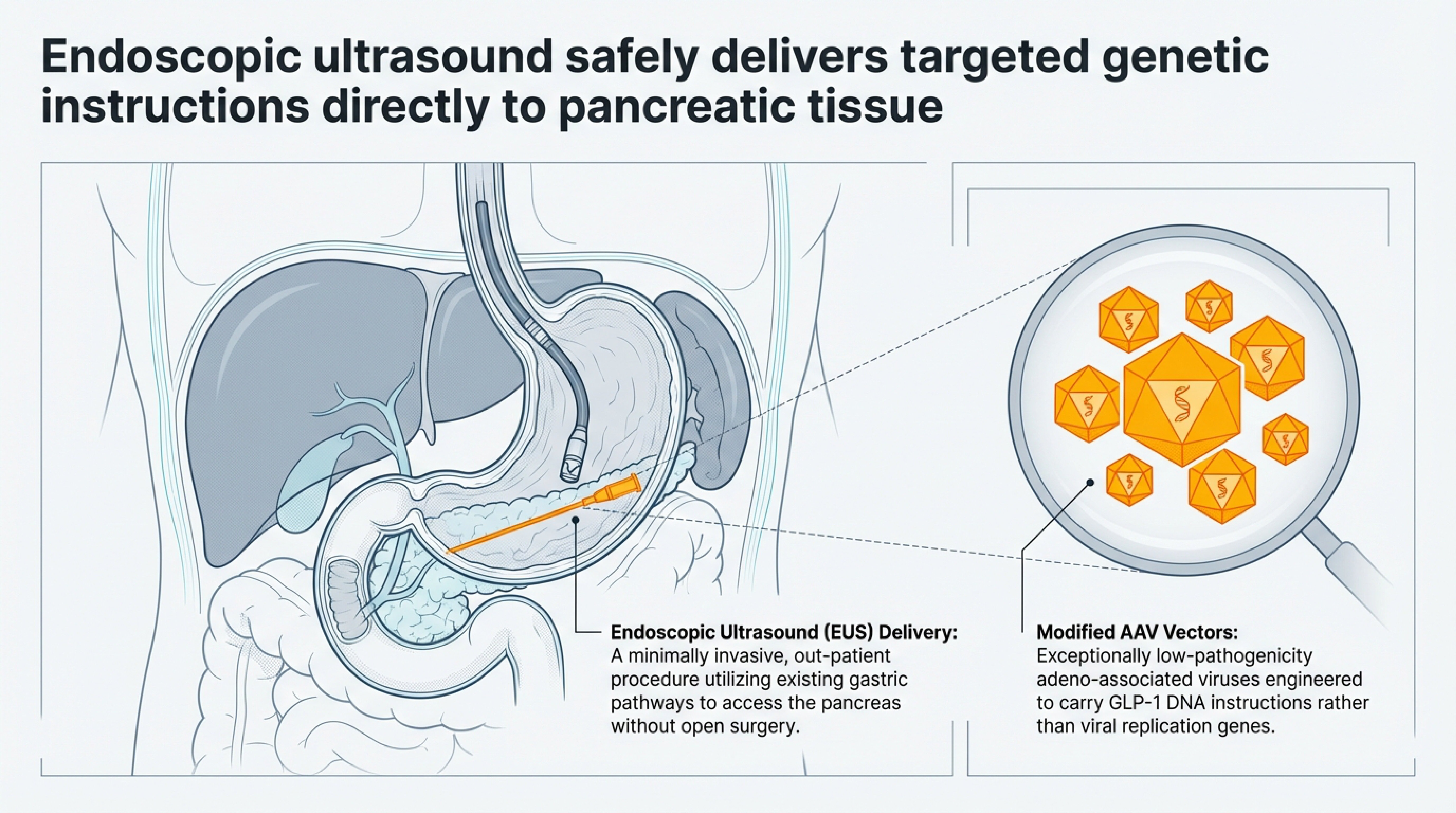
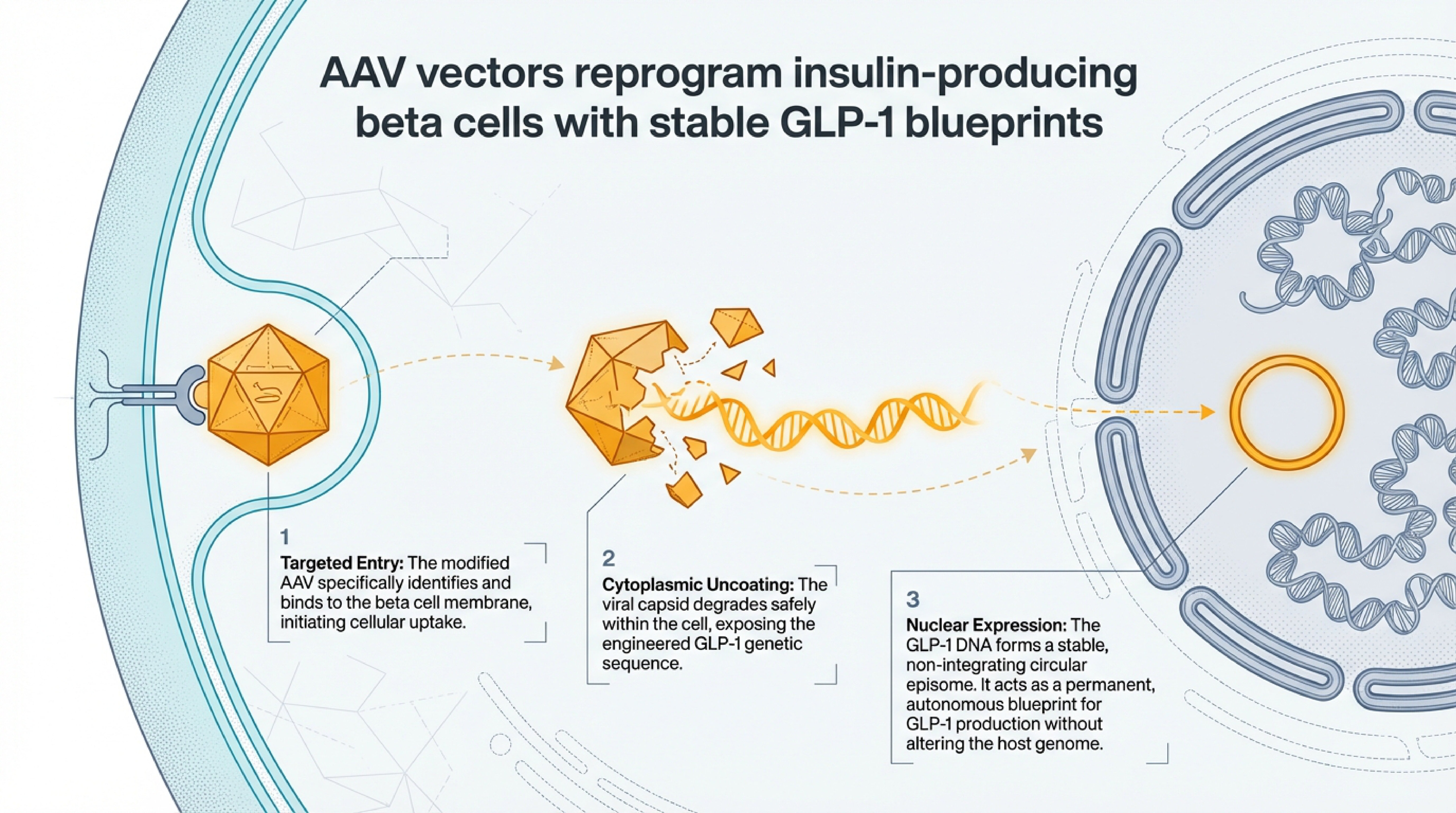
The delivery itself is something I find genuinely elegant as a physician who thinks a lot about minimally invasive procedures. As shown in the carousel above, Fractyl uses endoscopic ultrasound — a routine outpatient technique — to deliver their AAV vectors directly into pancreatic tissue. The AAV enters beta cells, uncoats, and forms a stable circular episome that acts as a permanent blueprint for GLP-1 production. No open surgery. No systemic viral exposure. The reprogrammed beta cells then co-secrete GLP-1 alongside insulin whenever they detect glucose — meal-triggered, just like your body's natural signaling.
Fractyl submitted its first Clinical Trial Application in Europe in May 2025. First-in-human dosing is anticipated in the second half of 2026. This is not a "someday" technology anymore. It's a regulatory milestone measured in months, not years.
Now, the interesting thing about Rejuva's design — and this matters clinically — is that it produces GLP-1 locally within pancreatic beta cells, triggered by nutrient sensing. It's not flooding your system with GLP-1 the way an injection does. At the ASGCT 2025 meeting, Fractyl presented data showing low systemic circulating GLP-1 levels with their approach. That's by design. If you can get the metabolic benefit without the systemic side effects — the nausea, the gastroparesis concerns — you've addressed one of the biggest barriers to long-term treatment.
The Independent Validation That Matters
One piece of evidence that doesn't get enough attention: in July 2025, a completely independent research group — not affiliated with Fractyl — published a peer-reviewed study in Nature's Communications Medicine demonstrating that a single genome-editing intervention targeting liver cells could produce sustained GLP-1 receptor agonist secretion. Their mice showed reduced food intake, weight gain attenuation, and improved insulin sensitivity with no detectable adverse effects.
This matters because the GLP-1 gene therapy concept is no longer supported by a single company's press releases alone. Independent researchers, using a different delivery mechanism and a different target tissue, arrived at the same conclusion: you can engineer cells to produce GLP-1 durably from a single treatment. The biological plausibility is peer-reviewed and replicated.
The Honest Risks — Because There Are Real Ones

I'll be direct about what we don't know, because I think patients deserve that. Gene therapy involves permanently modifying cellular DNA. AAV vectors are generally safe at therapeutic doses — eight AAV-based gene therapies have been FDA-approved as of 2024 — but at high doses, above roughly 10^14 viral genomes per kilogram, the literature documents immune reactions, liver toxicity, and in rare cases, genotoxicity.
Fractyl's approach specifically minimizes systemic exposure by targeting pancreatic beta cells and using a nutrient-responsive promoter rather than a constitutive one. That's a smart design decision. But smart design and long-term human safety data are two different things, and we don't have the latter yet.
The durability question is genuine. Mice live two to three years. We need these therapies to work for decades in humans. If expression wanes over time — which is possible — can you re-treat? AAV gene therapies sometimes trigger immune responses to the viral capsid that make repeat dosing difficult. This is an active area of research, not a solved problem.
And then there's the regulatory reality. Fractyl filed its CTA in Europe and Australia — not with the FDA. The U.S. regulatory path for GLP-1 gene therapy hasn't been formally engaged yet. The FDA did create a new Genetic Metabolic Diseases Advisory Committee in 2024, which expands its capacity to evaluate exactly this kind of therapy. But formal FDA guidance on gene therapy for obesity doesn't exist.
What This Means If You're a Patient Right Now
If you're currently on a GLP-1 drug and it's working for you, stay on it. These medications have established, proven benefits — cardiovascular protection, meaningful weight loss, improved glycemic control. The adherence challenge is real, but for the patients who tolerate the drugs well and can access them, the data is strong.
If you've tried GLP-1 drugs and stopped because of side effects or cost, know that the landscape is changing faster than most people realize. Oral formulations are in advanced development. Biosimilar competition is beginning to pressure pricing. And gene therapy approaches like Rejuva, if they succeed in human trials, could eventually offer a fundamentally different model — one treatment, sustained benefit, no weekly injections.
The most meaningful advances in medicine happen when we stop asking the body to depend on external interventions and start working with its own machinery. GLP-1 gene therapy is exactly that kind of thinking applied to metabolic disease.
Looking Forward
We're watching Fractyl's human trial data in 2026 with genuine scientific curiosity — not hype, but the kind of interest that comes from seeing independent validation, smart engineering, and a clear unmet need converge. The path from animal models to approved human therapy is long, and many promising programs fail along the way. But the question this research answers is no longer "is it biologically possible?" It is. The question now is whether it's safe, durable, and manufacturable at the scale that 100 million Americans with obesity would require.
That's a harder question. But it's the right one to be asking.