iPSC Therapy Restores Insulin Production: A Regenerative Approach to Type 1 Diabetes
🎧 Audio Overview
summary, or read the transcript below:
View Audio Transcript
A patient with Type 1 diabetes required no insulin injections for over a year following induced pluripotent stem cell therapy. This isn’t just remarkable—it demonstrates that regenerative medicine can address the underlying cellular deficit in Type 1 diabetes rather than merely managing symptoms.
Published in Nature in 2025, this case signals a fundamental shift. Instead of lifelong insulin replacement, we’re moving toward cellular replacement—restoring the body’s ability to produce insulin.
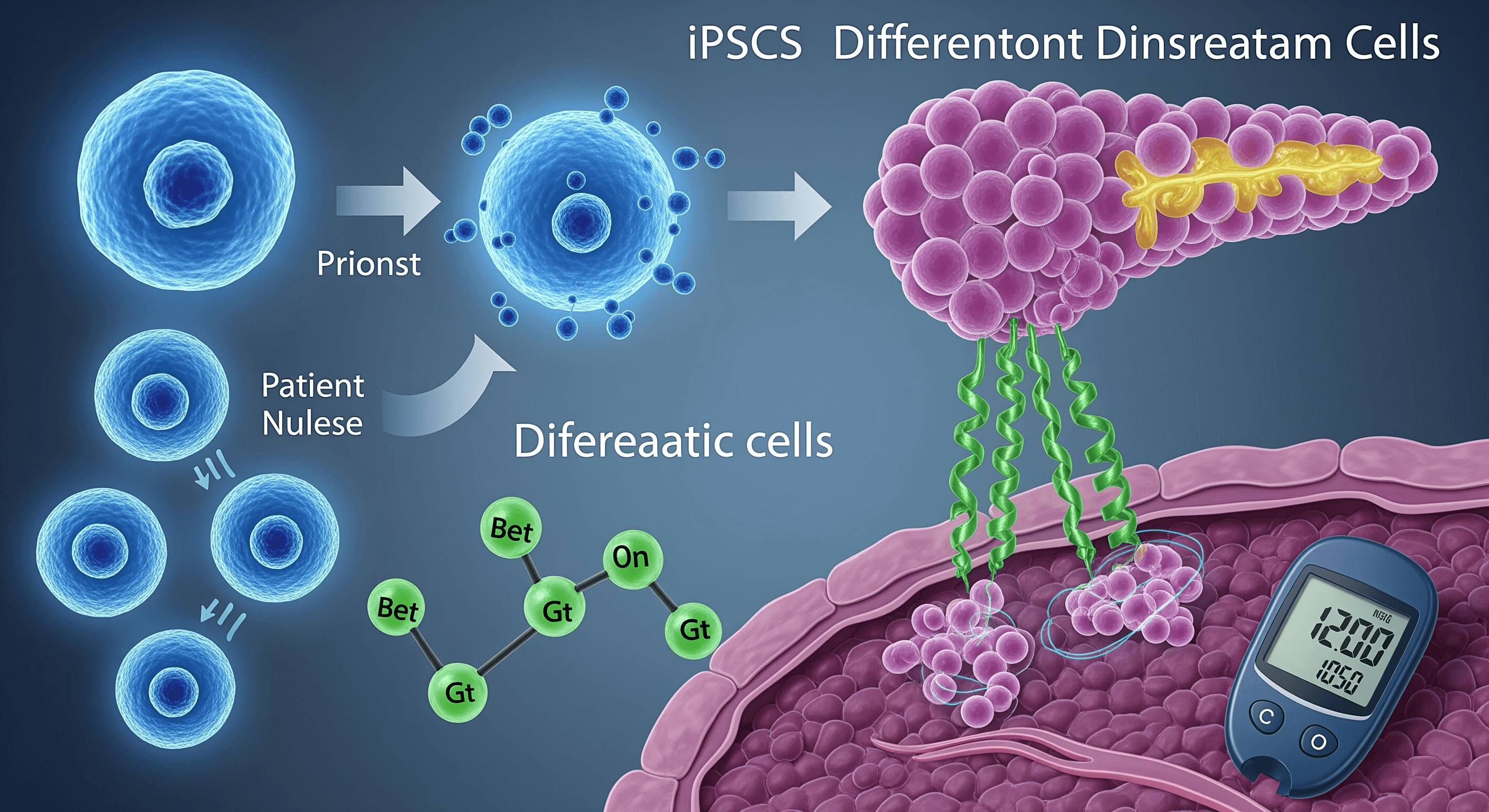
Researchers took the patient’s blood cells, reprogrammed them into iPSCs, then differentiated them into pancreatic beta cells—the insulin-producing cells destroyed by autoimmune attack in Type 1 diabetes.
These cells were transplanted into abdominal muscle. Within months, they began functioning. C-peptide levels rose. Insulin requirements declined. Eventually, the patient achieved insulin independence with normal glucose control.
More than a year post-transplant, she remained off insulin. Imaging showed viable, vascularized tissue. Biopsies confirmed functioning beta cells.
This represents the longest reported insulin independence following iPSC-derived beta cell transplantation in humans.
Challenges remain. Type 1 diabetes is autoimmune—new beta cells face the same immune attack as original ones. The patient still requires immunosuppression. Manufacturing is complex and expensive. Long-term safety data is limited.
But the principle is proven: patient-derived stem cells can be differentiated into functional insulin-producing cells that restore glucose regulation.
The question is no longer “Can we do this?” but “How do we optimize and scale this?”
For millions living with Type 1 diabetes, this offers realistic hope for cure, not just management.
A patient with Type 1 diabetes who required no insulin injections for over a year following induced pluripotent stem cell therapy represents more than a single remarkable case. It demonstrates proof-of-concept that regenerative medicine can address the underlying cellular deficit in Type 1 diabetes rather than merely managing its consequences.
Published in Nature in 2025, this case signals a fundamental shift in how we might approach autoimmune destruction of insulin-producing cells. Instead of lifelong insulin replacement, we may be moving toward cellular replacement—restoring the body’s ability to produce insulin endogenously.
The Challenge of Type 1 Diabetes
Type 1 diabetes results from autoimmune destruction of pancreatic beta cells—the specialized cells in the islets of Langerhans that produce insulin in response to blood glucose. Without functioning beta cells, patients cannot regulate blood sugar and require exogenous insulin administration.
Current management involves continuous glucose monitoring, insulin delivery (via multiple daily injections or insulin pumps), careful carbohydrate counting, and constant vigilance to avoid both hyperglycemia and hypoglycemia. Despite technological advances in glucose monitoring and insulin delivery, achieving optimal glucose control remains difficult, and many patients still develop complications including cardiovascular disease, kidney disease, neuropathy, and retinopathy.
The fundamental problem is that we’re replacing a hormone that should be produced endogenously in response to real-time glucose levels. Even the most sophisticated insulin pumps and continuous glucose monitors can’t perfectly replicate the second-to-second feedback system of functioning beta cells.
Regenerative approaches aim to restore that system by providing new beta cells that can sense glucose and secrete insulin appropriately.
Previous Approaches: Islet Transplantation
The concept of replacing beta cells isn’t new. Islet transplantation—transplanting islets from deceased donors into Type 1 diabetes patients—has been performed for over two decades following the Edmonton Protocol published in 2000.
Some islet transplant recipients achieved insulin independence for years, demonstrating that cellular replacement can work. But significant limitations prevented widespread adoption:
- Donor shortage: Each recipient requires islets from two or more deceased donors, making this approach impossible to scale
- Immunosuppression: Recipients require lifelong immunosuppressive drugs to prevent rejection, carrying their own significant risks and side effects
- Islet loss: Even with immunosuppression, transplanted islets often fail over time, requiring repeated transplants
- Autoimmune recurrence: The original autoimmune process that destroyed native beta cells can also attack transplanted cells
These challenges meant islet transplantation remained limited to select patients, typically those with severe hypoglycemia unawareness or those already requiring immunosuppression for kidney transplants.
Enter Induced Pluripotent Stem Cells
Induced pluripotent stem cells (iPSCs) are adult cells that have been reprogrammed to a pluripotent state—meaning they can differentiate into virtually any cell type in the body. The technology, developed by Shinya Yamanaka in 2006, earned him the Nobel Prize.
For Type 1 diabetes, the potential is clear: take a patient’s own cells, reprogram them to iPSCs, then differentiate them into insulin-producing beta cells. This approach theoretically solves multiple problems:
Unlimited supply: iPSCs can be expanded in culture, providing an essentially unlimited source of beta cells without relying on donor organs.
Autologous transplantation: Using a patient’s own cells should eliminate rejection risk, potentially reducing or eliminating need for immunosuppression.
Quality control: Lab-generated cells can be thoroughly characterized and tested before transplantation, ensuring they function properly and don’t carry unwanted cell types.
Genetic modification potential: Before differentiation, iPSCs could theoretically be genetically modified to resist autoimmune attack or improve function.
The challenge has been developing protocols to efficiently differentiate iPSCs into functional beta cells that appropriately sense glucose and secrete insulin. This requires recapitulating the complex developmental process that normally generates beta cells during pancreas formation.
The Nature Study: What Happened
The patient in this landmark case was a 25-year-old woman with long-standing Type 1 diabetes requiring substantial daily insulin. Researchers took her peripheral blood cells, reprogrammed them into iPSCs, then differentiated these into pancreatic islet cells using a multi-step protocol developed over years of research.
The differentiated cells were transplanted into the patient’s abdominal muscles—an accessible site that allows monitoring via imaging and biopsy if needed. This differs from traditional islet transplantation, which infuses cells into the portal vein where they lodge in the liver.
Within months, the transplanted cells began functioning. The patient’s C-peptide levels—a marker of endogenous insulin production—rose substantially. Her insulin requirements declined progressively. Eventually, she achieved insulin independence, maintaining normal glucose control without exogenous insulin.
More than a year post-transplant, she remained off insulin with excellent glucose control. Imaging showed the transplanted tissue was viable and vascularized. Biopsies confirmed the presence of functioning beta cells producing insulin.
This represents the longest reported period of insulin independence following iPSC-derived beta cell transplantation in a human patient.
What Makes This Work
Several factors appear critical to this success:
Differentiation Protocol
The researchers used a sophisticated multi-stage differentiation protocol that mimics the developmental stages pancreatic cells undergo during embryonic development. This process takes weeks and involves carefully timed exposure to specific growth factors and signaling molecules.
The resulting cells aren’t just insulin-producing cells—they’re organized into islet-like clusters containing multiple cell types (alpha, beta, delta cells) that normally populate pancreatic islets. This organization may be important for proper function.
Transplant Site
Using abdominal muscle rather than the liver offers several advantages. The site is accessible for monitoring and intervention if needed. It may provide a better microenvironment for survival and function. And it avoids the instant blood-mediated inflammatory reaction that can destroy islets infused into the portal vein.
Immunomodulation
While the patient received her own genetically matched cells, she still required immunosuppression—though the regimen appears less intensive than typically needed for allogeneic islet transplants. This addresses both rejection and recurrence of the autoimmune attack.
The need for immunosuppression despite using autologous cells highlights that the autoimmune process in Type 1 diabetes recognizes beta cell antigens, not foreign tissue antigens. New beta cells, even if genetically matched, still express the targets of autoimmune attack.
Cell Quantity and Quality
The transplant contained a substantial number of cells—enough to provide adequate insulin production even if not all cells survived. Quality control ensured the cells were properly differentiated, functional, and didn’t contain unwanted cell types that could cause complications.
Challenges and Limitations
While this case demonstrates feasibility, significant challenges remain before iPSC therapy becomes standard treatment for Type 1 diabetes.
The Autoimmune Problem
Type 1 diabetes is fundamentally an autoimmune disease. Unless we address the underlying immune dysfunction, newly transplanted beta cells face the same destruction as the original ones. Current approaches combine cellular replacement with immunosuppression, but this trades one treatment burden (insulin injections) for another (immunosuppressive drugs with their attendant risks).
Future solutions might include:
- Encapsulation devices that allow nutrients and insulin to pass while blocking immune cell access
- Genetic modification of iPSC-derived beta cells to make them “invisible” to the immune system
- Antigen-specific immunotherapies that selectively suppress the anti-beta cell response without broad immunosuppression
- Combination with immune reset strategies using immunoablation and immune reconstitution
Manufacturing and Scalability
The process of generating iPSC-derived beta cells takes months and requires sophisticated cell culture facilities. Scaling this to treat millions of Type 1 diabetes patients represents a massive manufacturing challenge.
Some research groups are developing “off-the-shelf” approaches using banked iPSC lines that match common HLA types, potentially allowing treatment without patient-specific cell generation. Others are creating hypoimmunogenic cells that evade immune recognition even with HLA mismatch.
Safety Considerations
Any stem cell therapy carries risks. Incompletely differentiated cells could form teratomas—tumors containing multiple tissue types. Cells that over-produce insulin could cause severe hypoglycemia. Quality control and safety monitoring are critical.
Long-term safety data remains limited. Will these cells continue functioning for decades? Will they maintain appropriate glucose-sensing and insulin secretion? Could they develop abnormalities over time?
Cost
Currently, iPSC therapy development is extraordinarily expensive. While costs should decrease with optimization and scale, this will likely remain a high-cost intervention, at least initially. Cost-effectiveness compared to lifelong insulin therapy and complication management will influence adoption.
Broader Context: Regenerative Diabetes Therapy
This iPSC work fits into a larger landscape of regenerative approaches to diabetes:
Stem cell-derived beta cells from embryonic stem cells: Similar to iPSCs but using embryonic rather than adult-derived stem cells. ViaCyte’s PEC-Encap product encapsulates such cells in an immune-protective device.
Beta cell regeneration: Attempts to stimulate remaining beta cells in patients with early Type 1 diabetes or Type 2 diabetes to proliferate and increase mass.
Transdifferentiation: Converting other pancreatic cell types (like alpha or delta cells) directly into beta cells without going through a stem cell stage.
Gene therapy: Using viral vectors to deliver genes that enable other cell types to produce insulin in response to glucose.
Each approach has strengths and limitations. iPSC therapy’s advantage lies in generating large numbers of patient-matched, well-characterized cells with proven function before transplantation.
Clinical Translation: What Comes Next
This Nature paper represents a single patient—an important proof-of-concept but not yet proven therapy. Clinical development will require:
Expanded clinical trials: Demonstrating reproducibility across multiple patients with diverse backgrounds, diabetes durations, and characteristics.
Longer follow-up: Proving durability beyond one year. Ideally, these cells would function for decades.
Optimization: Refining differentiation protocols, transplant sites, immunomodulation regimens, and patient selection criteria.
Regulatory approval: Navigating the complex regulatory pathway for cell therapy products requires extensive safety and efficacy data.
Manufacturing scale-up: Moving from research laboratory production to clinical-scale manufacturing with appropriate quality control.
Several companies and research groups are pursuing parallel development programs. Competition and collaboration in this space should accelerate progress.
Who Might Benefit First
Initial clinical applications will likely target patients with greatest unmet need:
- Those with severe hypoglycemia unawareness despite optimal insulin management
- Patients with extreme glucose variability that cannot be controlled with current technology
- Individuals already on immunosuppression for other reasons (kidney transplant recipients)
- Those willing to accept experimental therapy risk for potential cure
As safety and efficacy are established, indications could expand to broader Type 1 diabetes populations and potentially to patients with advanced Type 2 diabetes who have lost substantial beta cell function.
Implications for Regenerative Medicine
Beyond diabetes specifically, this work demonstrates that iPSC technology can generate complex, functional cell types that integrate into the body and perform sophisticated physiological tasks.
The principles being developed—cell differentiation protocols, transplant site selection, immunomodulation strategies, quality control methods—apply broadly to regenerative medicine. Success in diabetes could accelerate iPSC applications for other conditions: Parkinson’s disease, macular degeneration, heart failure, liver disease, and countless other conditions characterized by loss of specific cell types.
We’re witnessing the maturation of regenerative medicine from concept to clinical reality. The diabetes field is leading this transition in part because the target cell type (beta cells) and endpoint (insulin independence) are well-defined, making success measurable and development focused.
The Bigger Picture
Type 1 diabetes affects roughly 1.6 million Americans and millions more globally. Current management, while improved, remains burdensome and imperfect. Complications continue to develop despite best efforts.
A curative or near-curative approach would transform lives. Imagine patients freed from constant glucose monitoring, insulin calculations, and worry about hypoglycemia. Imagine children with Type 1 diabetes able to live without the burden this disease imposes on them and their families.
That’s the promise this research points toward. We’re not there yet—significant challenges remain—but the path forward is clearer. Patient-derived stem cells can be differentiated into functional insulin-producing cells that can restore glucose regulation when transplanted.
The question is no longer “Can we do this?” but “How do we optimize and scale this to help the millions who need it?”
Current Recommendations
For patients with Type 1 diabetes, what does this research mean right now?
This is not yet available clinical therapy. These remain early-stage investigations requiring years of additional development before potential FDA approval and clinical availability.
But it does mean that regenerative approaches are advancing rapidly. Patients and families should:
- Maintain optimal glucose control with current therapy to preserve health while these treatments develop
- Stay informed about clinical trials in regenerative diabetes therapy that may offer participation opportunities
- Connect with major academic diabetes centers that are likely to participate in future trials
- Support research funding for regenerative medicine and diabetes cure efforts
For those interested in the broader context of regenerative medicine and how cellular therapies are transforming treatment of chronic diseases, comprehensive discussions can be found in resources exploring advanced regenerative approaches and longevity science.
Looking Ahead
This single patient achieving insulin independence for over a year following iPSC-derived beta cell transplantation represents a milestone. It validates years of basic science research and brings regenerative therapy for Type 1 diabetes substantially closer to reality.
The next decade will likely see this approach move from proof-of-concept to established therapy, initially for select patients, then potentially for broader populations. Parallel advances in immunomodulation, encapsulation technology, and cell engineering will address current limitations.
We may be witnessing the beginning of the end of lifelong insulin dependence for Type 1 diabetes. That would represent one of regenerative medicine’s most significant achievements and provide a model for addressing other diseases through cellular replacement.
For the millions living with Type 1 diabetes, this research offers something that has been elusive: realistic hope for cure, not just management.
Dr. Pradeep Albert is a regenerative medicine physician and musculoskeletal radiologist specializing in advanced cellular therapies and longevity science. He is the author of “Exosomes, PRP, and Stem Cells in Musculoskeletal Medicine” and co-author of “Lifespan Decoded: How to Hack Your Biology for a Longer, Healthier Life.”